- Review article
- Open access
- Published:
Clinical Management of Nonsteroidal Anti-inflammatory Drug Hypersensitivity
World Allergy Organization Journal volume 1, pages 29–33 (2008)
Abstract
Hypersensitivity diseases caused by nonsteroidal anti-inflammatory agents are relatively common in the population. This article summarizes the present understanding on the various allergic and nonallergic clinical pictures produced through hypersensitivity to these drugs using the pathogenic classification of hypersensitivity reactions recently proposed by the Nomenclature Committee of the World Allergy Organization to guide clinicians in the diagnosis and management of patients with these conditions.
A large proportion of the population is exposed to nonsteroidal anti-inflammatory drugs (NSAIDs) worldwide from either medical prescription or self-medicated [1]. It is then not surprising that these drugs constitute the second major cause of hypersensitivity reactions to drugs after β-lactamic antibiotics.
The prevalence of these reactions in the population varies between 0.1% and 0.3%, [2] and therefore, it is very important for clinicians to recognize and properly treat patients suffering from NSAID hypersensitivity. This article reviews the information presently available on the clinical manifestations, diagnosis, and management of these reactions.
Nonsteroidal Anti-inflammatory Drugs
Pharmacology textbooks define NSAIDs as compounds that antagonize inflammation through the inhibition of a group of enzymes known as cyclooxygenases (COXs) [3]. Some drugs, notably pyrazolones and acetaminophen, were previously not classified into this group because they did not inhibit COX enzymes. In recent years, new COX isoenzymes have been described, such as COX-2b and COX-3, that can be selectively antagonized by these drugs, and therefore they would fit into the NSAID category [4, 5].
Classic NSAIDs that inhibit both major COX isoen-zymes, COX-1 and COX-2, can be classified according to their chemical structure as depicted in Table 1. A second classification is based on the selectivity of NSAIDs for inhibition of COX isoenzymes (Table 2).
Clinical Spectrum and Pathogenesis
A wide variety of clinical manifestations can be produced by NSAIDs. Using the classification proposed by the Nomenclature Committee of the World Allergy Organization, [6] the following types of hypersensitivity reactions can be considered:
Allergic Hypersensitivity
Immunologic reactions to NSAIDs can be subdivided into immediate (mediated by immunoglobulin E [IgE]) and delayed (mediated by lymphocytes).
Immediate Reactions
Urticaria and Angioedema
Immunoglobulin E-mediated cutaneous reactions have been described for pyrazolones, [7] acetaminophen, [8] and aspirin [9].
Allergic Anaphylaxis
Reported for ibuprofen, [10] ketorolac, [11] indomethacin, sulindac, zomepirac, [12] fenoprofen, meclofenamate, naproxen, piroxicam, tolmetin, [13] glafenine, acetaminophen, aspirin, diclofenac, and celecoxib [14].
Delayed Reactions
These include cell (T lymphocyte)-mediated type IV hypersensitivity reactions involving specific organs and systems.
Skin Diseases
Fixed-drug Eruptions
Characterized by erythematous plaques recurring in the same anatomical site in every occasion the drug is administered. Metamizole, piroxicam, phenylbutazone, paracetamol, aspirin, mefenamic acid, diclofenac, indomethacin, ibuprofen, diflunisal, naproxen, and nimesulide have been incriminated [15].
Toxic Epidermal Necrolysis, Stevens-Johnson Syndrome, and Acute Generalized Exanthyematous Pustulosis (AGEP)
These serious skin reactions belong to the erythema multiforme spectrum of bullous eruptions and can be associated with NSAIDs [16].
Stevens-Johnson syndrome (SJS) is a severe diffuse mucocutaneous eruption causing erythematous or purpuric macules, blisters, or target lesions with no more than 10% skin detachment, accompanied by systemic manifestations, occurring 1 to 8 weeks after administration of incriminated medications [17]. Toxic epidermal necrolysis (TEN) involves 30% or more skin detachment, whereas between 10% and 30% detachment is applied to the term SJS-TEN overlap syndrome.
Among NSAIDs, oxicams, phenylbutazone, and oxyphenbutazone have been responsible more often [16, 18]. Recently, a great deal of attention has been given to the association of SJS/TEN with the use of new COX-2 inhibitors, especially valdecoxib and celecoxib [19–21].
Acute generalized exanthematous pustulosis is a rare condition characterized by a rapid-onset pustular eruption involving most of the body. Typical lesions are generalized, nonfollicular, pinhead-sized sterile pustules on an erythematous background that are associated with fever and neutrophilia [22]. Histopathologic features include papillary edema, a mixed upper dermal perivascular infiltrate, and a spongiform subcorneal pustule. Activated HLA-DR-positive CD4 and CD8 T cells, interleukin-8, interleukin-5, and granulocyte-macrophage colony-stimulating factor are present in the tissue. The NSAIDs associated with acute generalized exanthematous pustulosis more often are ibuprofen, phenylbutazone, naproxen, acetylsalicylic acid, valdecoxib, and celecoxib.
Contact and Photocontact Dermatitis
Contact with NSAIDs can induce itchy, erythematous, edematous, and vesicular lesions, and photocontact dermatitis, an exaggerated or abnormal cutaneous response to light. Among NSAIDs, diclofenac, indomethacin, flurbiprofen, bufexamac, etofenamate, flufenamic acid, ibuprofen, ketoprofen, and tiaprofenic acid are the most common inducers of contact dermatitis. Cross-reactivity between some chemically related NSAIDs has been observed [23].
Maculopapular Eruptions
Virtually all NSAIDs are able to produce maculopapular eruptions, one of the most common cutaneous adverse effects of NSAIDs. Ibuprofen, pyrazolones, flurbiprofen, diclofenac, and celecoxib have been more frequently involved.
Pneumonitis
Some NSAIDs such as aspirin, sulindac, ibuprofen, and naproxen can induce allergic pneumonitis. The NSAID-induced pneumonitis can be suspected from a temporal relationship between lung infiltrates and drug administration [24]. Most patients will improve after drug discontinuation, although corticosteroids may be needed for severe or persistent cases.
Aseptic Meningitis
The NSAIDs are the medications more often involved in the production of drug-induced meningitis. Clinical features include fever, headache, photophobia, neck stiffness, nausea, vomiting, arthralgia, myalgia, rash, and abdominal pain [25]. Ibuprofen, sulindac, naproxen, tolmetin, diclofenac, ketoprofen, piroxicam, indomethacin, rofecoxib, and celecoxib have been associated with aseptic meningitis. Casas-Rodriguez et al [26] observed that 61% of ibuprofen-related meningitis occurred in patients with connective tissue diseases, mainly systemic lupus erythematosus. Management includes drug withdrawal, systemic corticosteroids, and avoidance of re-exposure to drugs from the same family as the causal drug.
Nephritis
Rarely, in aged patients with normal kidneys, NSAIDs may trigger a spectrum of nephritides ("NSAID nephropathy"), including tubular, interstitial, acute or subacute tubulointerstitial nephritis, chronic interstitial nephritis with papillary necrosis, and tubulointerstitial nephritis combined with nephrotic syndrome. The NSAIDs may also produce glomerulopathies such as minimal change nephropathy, membranous glomerulonephritis, and focal sclerosis [27, 28].
Hepatitis
Rarely, NSAIDs, among them niflumic acid, tolfenamic acid, diclofenac, fenoprofen, ibuprofen, indomethacin, naproxen, piroxicam, pirprofen, and sulindac, induce allergic hepatitis that can be mixed, cytolytic, or cholestatic. It is observed in elderly women taking multiple medications [29].
Herdeg et al [30] reported a case of metamizole-induced allergic cholestatic hepatitis characterized by generalized exanthema and increased liver enzymes. Sensitization to the drug was confirmed by means of lymphocyte transformation test.
Nonallergic Hypersensitivity
Composed of manifestations at the respiratory tract and skin, and nonallergic anaphylaxis.
Respiratory Hypersensitivity
Aspirin-induced asthma, aspirin-intolerant asthma, or aspirin-exacerbated respiratory disease (AERD) is characterized by asthma, rhinosinusitis, nasal polyposis, and aspirin/NSAID hypersensitivity. Asthmatic reactions induced by NSAIDs occur in 5% to 20% of adult asthmatic patients [31]. The pathogenesis seems to involve the combined effects of chronic inflammation and a pharmacogenetic abnormality of arachidonic acid metabolism in response to NSAIDs. This leads to sulfidoleukotriene overproduction and to a decrease in anti-inflammatory prostaglandin E2 from insufficient COX-2 activation, leading to additional leukotriene synthesis [32, 33]. Excellent reviews on this topic have been recently published [34].
Cutaneous Hypersensitivity
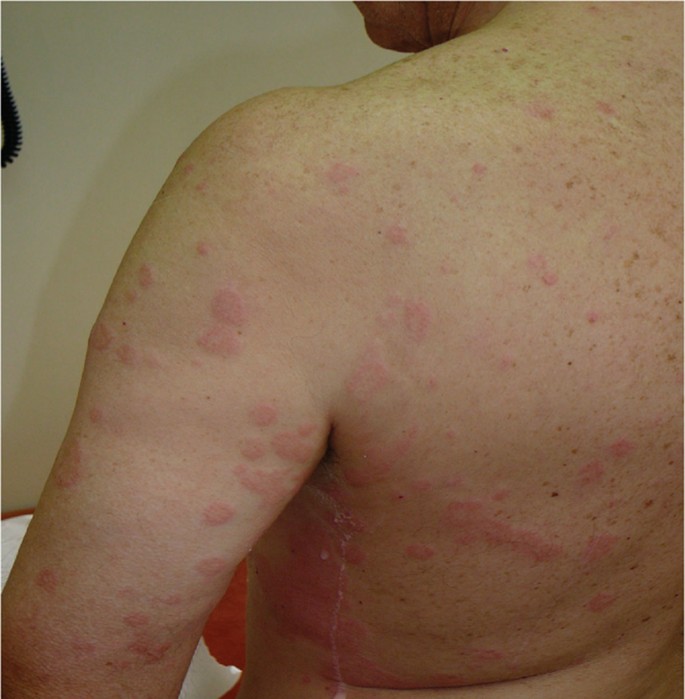
The cutaneous pattern of NSAID-induced cross-reactions includes cross-reacting urticaria and angioedema in patients with or without chronic idiopathic urticaria (CIU) (Figure 1). The mechanisms are not completely understood, but in patients with CIU, COX-1 inhibition has been demonstrated [35, 36].
Nonallergic Anaphylaxis
Previously known as anaphylactoid or pseudoallergic reaction, it is observed in cross-reactive patients and presumably mediated by inhibition of COX-1 [37].
Diagnostic Methodology for Adverse Reactions Induced by NSAIDS
The choice of diagnostic tests is based on the clinical picture and possible pathogenesis.
Reactions Mediated by IgE
Although intradermal injection of pyrazolones has been proposed for diagnostic purposes, no correlation with the clinical picture was observed [7]. Presently, no standardized reagents for immediate-type skin tests with NSAIDs are available.
Delayed-type Reactions
Patch tests constitute a simple, fast, and relatively safe method for the diagnosis of delayed reactions to NSAIDs. For fixed-drug eruptions, lesional skin should be used for the test [38–40]. When photoallergy is suspected, photopatch tests are indicated [41]. Concentrations of NSAIDs commonly used for patch tests are summarized in Table 3.
It must be noticed that in some cases, a rechallenge with the drug is not recommended because of the high risk of severe and generalized reactions [42]. Intradermal and scratch tests with reading at 48 and 72 hours have also been used to confirm delayed hypersensitivity to NSAIDs [43].
The lymphocyte transformation test measures the in vitro proliferative response of T cells stimulated by the drug. Although this test is worth to be considered, it is only available in some laboratory facilities in specialized centers [44].
Nonallergic Reactions
For respiratory and cutaneous cross-reactions, the criterion standard continues to be the controlled oral provocation test carried out in the appropriate medical facilities by physicians experienced in this kind of test and with easy access to medications and equipments necessary for the treatment of reactions [45, 46]. Bronchial and nasal inhalation challenges with aerosols of L-lysine acetylsalicylic acid have also been used [47, 48]. An algorithm for the diagnosis and management of patients with cutaneous NSAID reactions has been proposed [46].
De Weck et al [49] have developed 2 in vitro assays done with blood basophils: the leukotriene release test and the basophil activation test. These tests require, however, special equipments and reagents, including a flow cytometer, and therefore are more expensive and limited to some centers [50].
Patient Management
IgE-mediated Reactions
Patients reacting to a single NSAID can receive NSAIDs from a different chemical group (see Table 1). In general, it is recommended not to use in these patients NSAIDs from the same group because cross-reactions between NSAIDs of similar chemical structure occur.
Delayed-type Reactions
Discontinuation of offender medication and pharmacological treatment with corticosteroids and antihistamines are recommended.
Patients with severe reactions of the TEN/SJS type should be transferred to intensive care or burn units. Intravenous immunoglobulins have been used in patients with TEN, based on its content of natural anti-Fas antibodies, and a reduction of mortality has been shown in some studies [51]. Systemic therapy with infliximab (anti-TNF-α) induced rapid improvement of skin lesions in patients with TEN [52].
Nonallergic Reactions
Aspirin-exacerbated Respiratory Disease
The following measures are recommended for patients with AERD:
-
Avoidance of all classic COX-1 inhibitors.
-
Pharmacological treatment with topical and systemic corticosteroids, leukotriene receptor antagonists, and 5-lipoxygenase inhibitors, antibacterials, and antifungals [53].
-
Use of alternative NSAIDs (acetaminophen, salsalate, dextropropoxyphene, opioids, ergotamine, hyoscine, sodium salicylate, salicylamide, choline-magnesium trisalicylate, floctafenine). Acetaminophen and other weak inhibitors of COX-1 are generally well tolerated by these patients at lower doses, but if the dose is increased, respiratory reactions can occur [54].
-
Specific COX-2 inhibitors are tolerated by most patients with AERD [55].
-
Desensitization is indicated for selected sensitive patients who need to receive NSAIDs for other medical conditions and for patients with severe corticosteroid-dependent AERD [56].
Cutaneous Reactions and Nonallergic Anaphylaxis
Patients with cross-reacting urticaria/angioedema and nonallergic anaphylaxis should be managed as follows:
-
Avoidance of COX-1 inhibitors.
-
Alternative medications as mentioned above (see aspirin-exacerbated respiratory disease).
-
COX-2 inhibitors are safe for most of these patients, but long-term use of coxibs is not recommended because of the cardiovascular risks associated with them. In such cases, preferential inhibitors of COX-2 may be helpful [57, 58].
-
Desensitization is generally not recommended.
References
Sánchez-Borges M, Capriles-Hulett A, Caballero-Fonseca F: Risk of skin reactions when using ibuprofen-based medicines. Expert Opin Drug Saf. 2005, 4: 837-848. 10.1517/14740338.4.5.837.
Settipane GA: Aspirin and allergic diseases. Am J Med. 1983, 74: 102-110.
Goodman and Gillman's. The Pharmacological Basis of Therapeutics. Edited by: Brunton L, Lazo J, Parker K. 2005, New York, NY: McGraw-Hill Publishing, 11
Chandrasekharan NV, Dai H, Roos KL, et al: COX-3, a cyclooxygenase-1 variant inhibited by acetaminophen and other analgesic/antipyretic drugs: cloning, structure and expression. Proc Natl Acad Sci USA. 2002, 99: 13926-13931. 10.1073/pnas.162468699.
Simmons DL, Botting RM, Robertson PM, et al: Induction of an acetaminophen-sensitive cyclooxygenase with reduced sensitivity to nonsteroidal anti-inflammatory drugs. Proc Natl Acad Sci USA. 1999, 96: 3275-3280. 10.1073/pnas.96.6.3275.
Johansson SGO, Bieber T, Dahl R, et al: Revised nomenclature for allergy for global use: report of the Nomenclature Review Committee of the World Allergy Organization, October 2003. J Allergy Clin Immunol. 2004, 113: 832-836. 10.1016/j.jaci.2003.12.591.
Kowalski ML, Bienkiewicz B, Woszczek G: Diagnosis of pyrazolone drug sensitivity: clinical history versus skin testing and in vitro testing. Allergy Asthma Proc. 1999, 20: 347-352. 10.2500/108854199778251799.
Grant JA, Weiler JM: A report of a rare immediate reaction after ingestion of acetaminophen. Ann Allergy Asthma Immunol. 2001, 87: 227-229. 10.1016/S1081-1206(10)62231-4.
Blanca M, Perez E, Garcia JJ, et al: Angioedema and IgE antibodies to aspirin: a case report. Ann Allergy. 1989, 62: 295-298.
Takahama H, Kubota Y, Mizoguchi M: A case of anaphylaxis due to ibuprofen. J Dermatol. 2000, 27: 337-340.
Scala E, Giani M, Pirrotta L, et al: Selective severe anaphylactic reaction due to ketorolac tromethamine without nonsteroidal anti-inflammatory drug intolerance. J Allergy Clin Immunol. 2001, 107: 557-10.1067/mai.2001.113241.
Strom BL, Carson JL, Lee Morse M, et al: The effect of indication on hypersensitivity reactions associated with zomepirac sodium and other nonsteroidal anti-inflammatory drugs. Arthritis Rheum. 1987, 30: 1142-1148. 10.1002/art.1780301009.
Strom BL, Carson JL, Schinnar R: The effect of indication on the risk of hypersensitivity reactions associated with tolmetin sodium versus other nonsteroidal anti-inflammatory drugs. J Rheumatol. 1988, 15: 695-699.
Levy MB, Fink JN: Anaphylaxis to celecoxib. Ann Allergy Asthma Immunol. 2001, 87: 72-73. 10.1016/S1081-1206(10)62327-7.
Mahboob A, Saeed Haroon T: Drugs causing fixed eruptions. A study of 450 cases. Int J Dermatol. 1998, 37: 833-838. 10.1046/j.1365-4362.1998.00451.x.
Mockenhaupt M, Kelly JP, Kaufman D, Stern RS, SCAR Study Group: The risk of Stevens-Johnson syndrome and toxic epidermal necrolysis associated with nonsteroidal anti-inflammatory drugs: a multinational perspective. J Rheumatol. 2003, 30: 2234-2240.
Lyell A: Toxic epidermal necrolysis (the scalded skin syndrome): a reappraisal. Br J Dermatol. 1979, 100: 69-86. 10.1111/j.1365-2133.1979.tb03571.x.
Guillaume JC, Roujeau JC, Revuz J, et al: The culprit drugs in 87 cases of toxic epidermal necrolysis (Lyell's syndrome). Arch Dermatol. 1987, 123: 1166-1170. 10.1001/archderm.1987.01660330077013.
Layton D, Marshall V, Boshier A, et al: Serious skin reactions and selective COX-2 inhibitors. A case series from prescription-event monitoring in England. Drug Saf. 2006, 29: 687-696. 10.2165/00002018-200629080-00005.
La Grenade L, Lee L, Weaver J, et al: Comparison of reporting of Stevens-Johnson syndrome and toxic epidermal necrolysis in association with selective COX-2 inhibitors. Drug Saf. 2005, 28: 917-924. 10.2165/00002018-200528100-00008.
Sánchez-Borges M, Capriles-Hulett A, Caballero-Fonseca F: Adverse reactions to selective cyclooxygenase-2 inhibitors (coxibs). Am J Ther. 2004, 11: 494-500. 10.1097/01.mjt.0000125121.35422.b4.
Roujeau JC, Bioulac-Sage P, Bourseau C, et al: Acute generalized exanthematous pustulosis. Analysis of 63 cases. Arch Dermatol. 1991, 127: 1333-1338. 10.1001/archderm.1991.01680080069004.
Pigatto PD, Mozzanica N, Bigardi AS, et al: Topical NSAID allergic contact dermatitis--Italian experience. Contact Dermatitis. 1993, 29: 39-40.
Allen JN: Drug-induced eosinophilic lung disease. Clin Chest Med. 2004, 25: 77-88. 10.1016/S0272-5231(03)00141-2.
Jolles S, Sewell C, Leighton C: Drug-induced aseptic meningitis. Diagnosis and management. Drug Saf. 2000, 22: 215-226. 10.2165/00002018-200022030-00005.
Casas-Rodriguez S, Manzur Olguin A, Peña Miralles C, et al: Characteristics of meningitis caused by ibuprofen. Report of 2 cases with recurrent episodes and review of the literature. Medicine. 2006, 85: 214-220. 10.1097/01.md.0000229757.78057.50.
Ravnskov U: Glomerular, tubular and interstitial nephritis associated with non-steroidal anti-inflammatory drugs. Evidence of a common mechanism. Br J Clin Pharmacol. 1999, 47: 203-210.
Brezin JH, Katz SM, Schwartz AB, Chiwitz JL: Reversible renal failure and nephrotic syndrome associated with nonsteroidal anti-inflammatory drugs. N Engl J Med. 1979, 301: 1271-1273. 10.1056/NEJM197912063012306.
Hannequin JR, Doffoel M, Schmutz G: Rev Rheum Mal Osteoartic. 1988, 55: 983-988. [in French]
Herdeg C, Hilt F, Büchtemann A, et al: Allergic cholestatic hepatitis and exanthema induced by metamizole: verification by lymphocyte transformation test. Liver. 2002, 22: 507-513. 10.1034/j.1600-0676.2002.01718.x.
Sánchez-Borges M, Capriles-Hulett A, Caballero-Fonseca F: NSAID hypersensitivity in the COX-2 inhibitor era. Allergy Clin Immunol Int. 2001, 13: 211-217. 10.1027/0838-1925.13.5.211.
De Weck A, Sanz ML, Gamboa P: New pathophysiological concepts on aspirin hypersensitivity (Widal syndrome); diagnostic and therapeutic consequences. Bull Acad Natl Med. 2005, 189: 1201-1218.
Picado C: The role of cyclooxygenase in acetylsalicylic acid sensitivity. Allergy Clin Immunol Int: J World Allergy Org. 2006, 18: 154-157. 10.1027/0838-1925.18.4.154.
Kowalski ML, Makowska JS: Aspirin-exacerbated respiratory disease. An update on diagnosis and management. Allergy Clin Immunol Int: J World Allergy Org. 2006, 18: 140-149. 10.1027/0838-1925.18.4.140.
Zembowicz A, Mastalerz L, Setkowicz M, et al: Safety of cyclo-oxygenase-2 inhibitors and increased leukotriene synthesis in chronic idiopathic urticaria with sensitivity to non steroidal anti-inflammatory drugs. Arch Dermatol. 2003, 139: 1577-1582. 10.1001/archderm.139.12.1577.
Sánchez-Borges M, Capriles-Hulett A, Caballero-Fonseca F: The multiple faces of nonsteroidal anti-inflammatory drug hypersensitivity. J Investig Allergol Clin Immunol. 2004, 14: 329-334.
Berkes EA: Anaphylactic and anaphylactoid reactions to aspirin and other NSAIDs. Clin Rev Allergy Immunol. 2003, 24: 137-148. 10.1385/CRIAI:24:2:137.
Zedlits S, Linzbach L, Kauffmann R, Boehncke WH: Reproducible identification of the causative drug of a fixed drug eruption by oral provocation and lesional patch testing. Contact Dermatitis. 2002, 46: 352-353. 10.1034/j.1600-0536.2002.460606.x.
Whittam LR, Wakelin SH, Barker JNWN: Generalized pustular psoriasis or drug-induced pustuloderma? The use of patch testing. Clin Exp Dermatol. 2000, 25: 122-124.
Alanko T, Stubb S, Reitamo S: Topical provocation of fixed drug eruption. Br J Dermatol. 1987, 116: 561-567. 10.1111/j.1365-2133.1987.tb05879.x.
Hasan T, Jansen CT: Photopatch test reactivity: effect of photoallergen concentration and UVA dosing. Contact Dermatitis. 1996, 34: 383-386. 10.1111/j.1600-0536.1996.tb02237.x.
Kauppinen K, Stubb S: Fixed eruptions. Causative drugs and challenge tests. Br J Dermatol. 1985, 112: 575-578.
Talhari C, Lauceviciute I, Enderlein E, et al: COX-2 selective inhibitor valdecoxib induces severe allergic skin reactions. J Allergy Clin Immunol. 2005, 115: 1089-1090. 10.1016/j.jaci.2004.12.1135.
Pichler WJ, Tilch J: The lymphocyte transformation test in the diagnosis of drug hypersensitivity. Allergy. 2004, 59: 809-820. 10.1111/j.1398-9995.2004.00547.x.
Fahrenholz JM: Natural history and clinical features of aspirin-exacerbated respiratory disease. Clin Rev Allergy Immunol. 2003, 24: 113-124. 10.1385/CRIAI:24:2:113.
Sánchez-Borges M, Capriles-Hulett A, Caballero-Fonseca F: Cutaneous reactions to aspirin and nonsteroidal anti-inflammatory drugs. Clin Rev Allergy Immunol. 2003, 24: 125-136. 10.1385/CRIAI:24:2:125.
Nizankowska E, Bestynska-Krypel A, Cmiel A, Szczeklik A: Oral and bronchial provocation test with aspirin for diagnosis of aspirin-induced asthma. Eur Respir J. 2000, 15: 863-869. 10.1034/j.1399-3003.2000.15e09.x.
Milewski M, Mastalerz L, Nizanskowska E, Szczeklik A: Nasal provocation test with lysine-aspirin for diagnosis of aspirin-sensitive asthma. J Allergy Clin Immunol. 1999, 101: 581-586.
De Weck AL, Gamboa PM, Esparza R, Sanz ML: Hypersensitivity to aspirin and other nonsteroidal anti-inflammatory drugs (NSAIDs). Curr Pharm Des. 2006, 12: 3347-3358. 10.2174/138161206778193971.
Sanz ML, Gamboa P, De Weck AL: A new combined test with flowcytometric basophil activation and determination of sulfidoleukotrienes is useful for in vitro diagnosis of hypersensitivity to aspirin and other nonsteroidal anti-inflammatory drugs. Int Arch Allergy Immunol. 2005, 136: 58-72. 10.1159/000082586.
French LE, Trent TJ, Kerdel FA: Use of intravenous immunoglobulin in toxic epidermal necrolysis and Stevens Johnson syndrome: our current understanding. Int Immunopharmacol. 2006, 6: 543-549. 10.1016/j.intimp.2005.11.012.
Hunger RE, Hunziker T, Buettiker U, et al: Rapid resolution of toxic epidermal necrolysis with anti-TNF-á treatment. J Allergy Clin Immunol. 2005, 116: 923-924. 10.1016/j.jaci.2005.06.029.
Stevenson DD, Simon RA: Selection of patients for aspirin desensitization treatment. J Allergy Clin Immunol. 2006, 118: 801-804. 10.1016/j.jaci.2006.06.019.
Settipane RA, Stevenson DD: Cross sensitivity with acetaminophen in aspirin-sensitive subjects with asthma. J Allergy Clin Immunol. 1989, 84: 26-33. 10.1016/0091-6749(89)90174-7.
Kowalski ML, Makowska J: Use of nonsteroidal anti-inflammatory drugs in patients with aspirin hypersensitivity: safety of cyclo-oxygenase-2 inhibitors. Treat Respir Med. 2006, 5: 399-406. 10.2165/00151829-200605060-00005.
Berges-Gimeno MP, Simon RA, Stevenson DD: Long term desensitization with aspirin in aspirin-exacerbated respiratory disease. J Allergy Clin Immunol. 2003, 111: 180-186. 10.1067/mai.2003.7.
Sánchez-Borges M, Capriles-Hulett A, Caballero-Fonseca F: Cutaneous hypersensitivity reactions to inhibitors of cyclooxygenase-2. Results from 307 oral provocation tests and review of the literature. Allergy Clin Immunol Int: J World Allergy Org. 2007, 19: 44-49. 10.1027/0838-1925.19.2.44.
Sánchez-Borges M, Capriles-Hulett A, Caballero-Fonseca F: Crossreactive cutaneous hypersensitivity to nonsteroidal anti-inflammatory drugs. Are preferential cyclooxygenase-2 inhibitors a therapeutic option?. Allergy Clin Immunol Int: J World Allergy Org. 2007, 19: 91-94.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Sánchez-Borges, M. Clinical Management of Nonsteroidal Anti-inflammatory Drug Hypersensitivity. World Allergy Organ J 1, 29–33 (2008). https://doi.org/10.1097/WOX.0b013e3181625db2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1097/WOX.0b013e3181625db2